1. INTRODUCTION
Epilepsy is one of the most popular chronic neurological diseases and is characterized by epileptic seizures that cause neurobiological, psychological, cognitive, and social consequences [1]. This implies that epilepsy has long-lasting effects on the lifestyles of people with epilepsy (PWE) and their families; therefore, the quality of life has been significantly affected. Optimal management of epilepsy should include both medical and psychosocial aspects. Recent studies have focused on not only pharmacotherapy, but also on social impact of PWE. Quality of life is assessed as an outcome measure in clinical practice and trials. Several instruments were developed to measure quality of life in general, or with respect to specific diseases, including epilepsy. There are some quality-of-life tools for epilepsy, i.e., Liverpool Assessment Battery, the Quality of Life in Epilepsy-89 Inventory (QOLIE-89), the 31-item Quality of Life in Epilepsy Inventory (QOLIE-31), and the Quality of Life in Epilepsy-10 Inventory (QOLIE-10).
In Vietnam, the Health-related Quality of Life Measure for Children with Epilepsy (CHEQOL-25) was translated and validated [2]; but there is no specific instrument to assess the quality of life in adult PWE. Recognizing the need for such a scale, we conducted a translation and validation of quality-of-life instrument for PWE. The QOLIE-31 was initially developed and validated in the United States (US) [3]. It comprises seven subscales which covers both general and epilepsy-specific domains. We chose the QOLIE-31 due to several reasons. Firstly, QOLIE-31 was the most popular scale used in PWE. Thirty-one among 194 countries in the World Health Organization (WHO) with 7255 individuals were reported using this scale to assess quality of life in epilepsy [4]. Second, this instrument was translated and validated in different languages around the world; so it is uniform across languages and cultures [4]. The availability of a valid cross-cultural instrument is important for future international studies, and studies that may want to compare the results among various countries. Furthermore, QOLIE-31 is most suitable with enough length and simplicity compared with other tools. QOLIE-10 scale is used for scanning because of its shortage, Quality of Life in Epilepsy for Adolescents (QOLIE-AD-48) is used in a specific group, or Liverpool HRQOL scale and QOLIE-89 with multiple scales are rather complex to assess. This study aimed to translate and validate the QOLIE-31 measurement into Vietnamese.
2. MATERIALS AND METHOD
This cross-sectional study was conducted at the epilepsy clinic at Nguyen Tri Phuong Hospital, Ho Chi Minh City, Viet Nam.
To address the reliability and validity studies in neurology, the authors reported that the minimum sample sizes of 20 for reliability and 80 for validity allowed high representative of the main study samples [5]. And we chose convenience sampling for this current study.
The QOLIE-31 is a shortened version of the QOLIE-89 and comprises seven subscales including: seizure worry (SW), overall QOL (QOL), energy and fatigue (EF), cognitive function (CF), medication effects (ME), emotional well-being (EWB), and social functioning (SF). These subscales are grouped into two factors: Emotional, Psychological Effects (SW, QOL, EWB, and EF) and Medical/Social Effects (ME, CF, and SF). Subscale and total scores are calculated according to the participants’ answers. The scoring procedure for the QOLIE-31 includes converting the raw pre-coded numeric values of items to 0-100 scores, with higher scores reflecting a better quality of life. The Scoring Manual of the QOLIE-31 (version 1.0) guided the calculation of the total and subscale scores. (https://www.rand.org/content/dam/rand/www/external/health/surveys_tools/qolie/qolie31_scoring.pdf) [6]
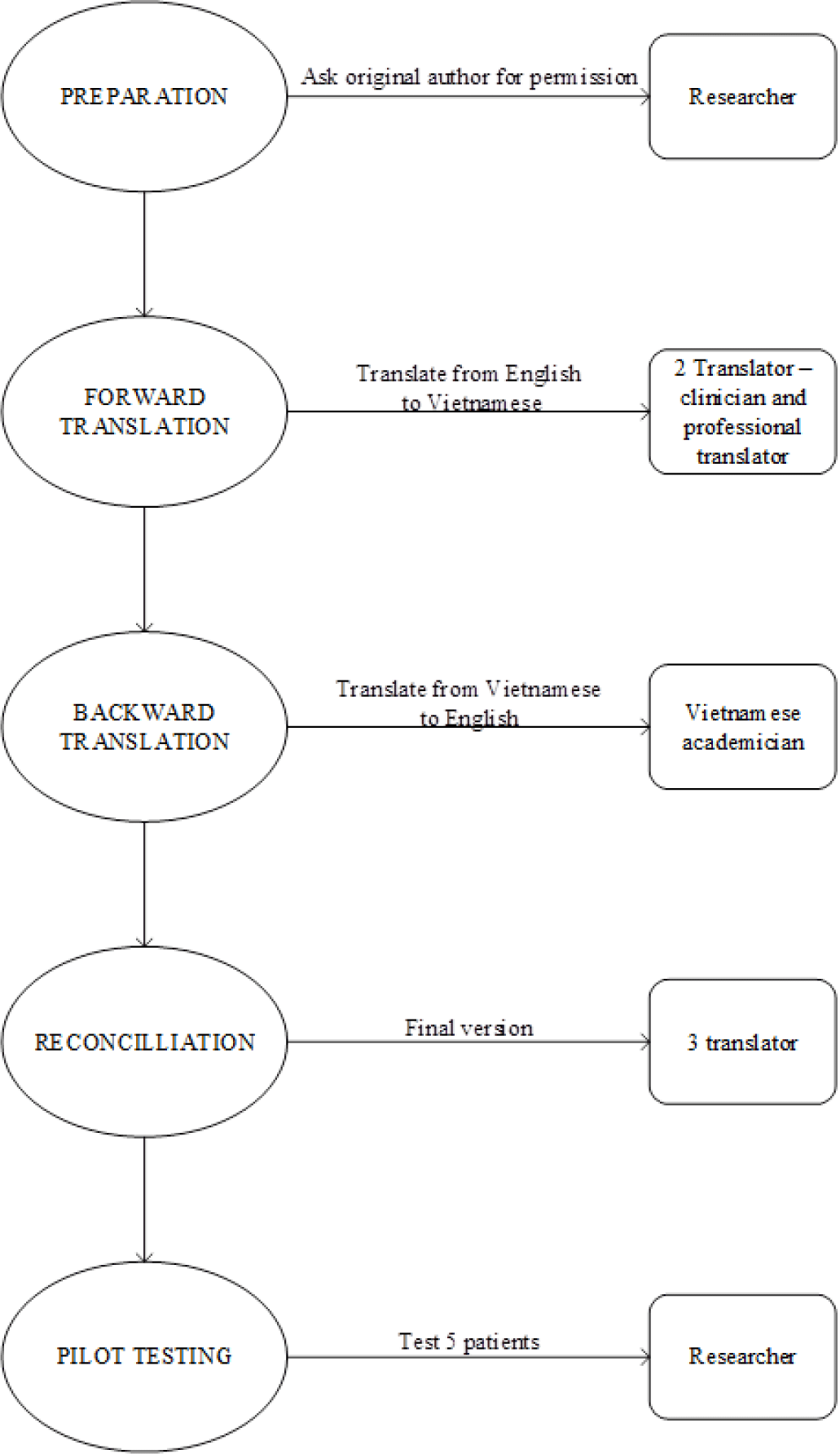
Prior to translating the inventory, permission was obtained via email from the developer of original instrument, Dr. Joyce Cramer. The translation and cross-cultural adaptation of the original version of the QOLIE-3 to Vietnamese followed the Principles of Good Practice for the Translation and Cultural Adaptation of Patient-Reported Outcomes Measures of International Society for Pharmacoeconomics and Outcomes Research (ISPOR). This included the stages of preparation, forward translation, reconciliation, back translation, back translation review, harmonization, cognitive debriefing, and finalization [7].
The first step was forward translation by two bilingual translators, one a clinician and the other, a professional translator. Both translations were then compared and discussed to create the combined version. The resulting inventory was then back translated into English by a Vietnamese academician with a master’s degree in English and no medical knowledge. The back-translated English version was sent to Dr. Joyce Cramer, who checked the conceptual equivalence of the translation. Three translators and one neurologist compared documents to define a final version that matched the intent and style of the original US English instrument as closely as possible. Cognitive debriefing was the next step to evaluate the translated instrument with five adult PWE. Participants were asked to judge the clarity of the content by reviewing the various items and stating whether they understand the item (Figure 1).
Owing to cultural and economic differences, we adjusted the content of item 20. In Vietnam, the most common means of transportation is driving a motorbike or bicycle, instead of a car. For this reason, item 20 was altered from “driving” in the original scale to “driving or using other transportation (e.g., motorbike, bicycle)” in the Vietnamese version.
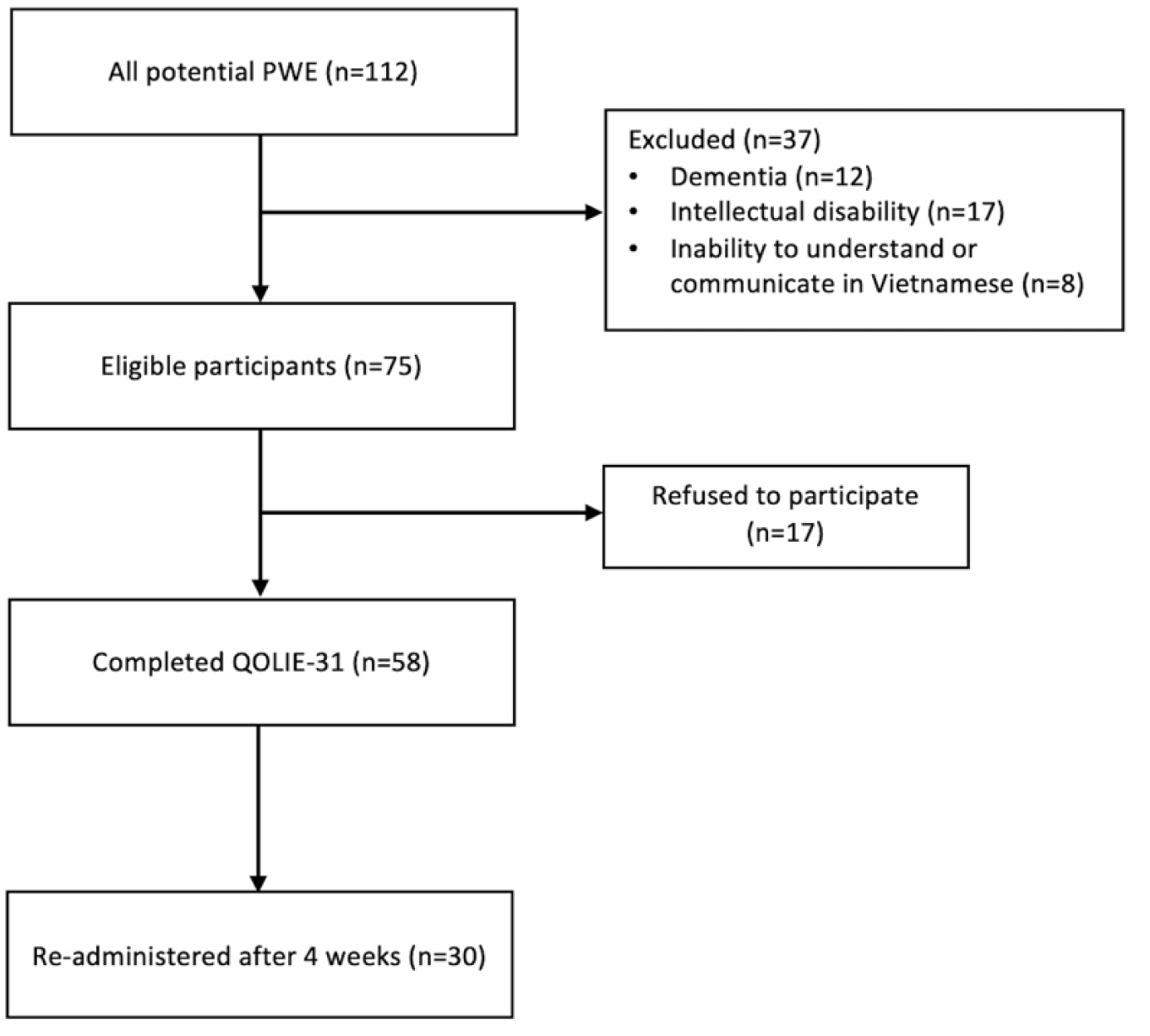
To assess the reliability and validity of the QOLIE-31, a sample of non-hospitalized PWE were chosen from the Epilepsy clinic at Nguyen Tri Phuong Hospital, Ho Chi Minh city in Viet Nam. Study participants were consecutively enrolled from June 2020 to September 2020. Participants were eligible if they were (1) above 18 years old, and (2) diagnosed with epilepsy according to the requirements of The International League Against Epilepsy (ILAE) [1]. The exclusion criteria were: (1) people with an intellectual disability, learning disability, cognitive impairment or (2) limited Vietnamese proficiency, which may hinder the PWE in completing the questionnaire. All eligible participants received a detailed explanation of the background of the study and were required to provide written informed consent before study enrollment.
The investigators interviewed face-to-face participants to collect demographic and clinical information including gender, age, educational levels, employment, marital status, seizure types, etiology, duration of illness, seizure outcome, and the number of antiepileptic drugs (AEDs). Seizure outcome included the following variables: seizure-free from 1 year, drug-resistant defined by ILAE [8], and undetermined with insufficient information to be drug-resistant. Subsequently, the participants completed the QOLIE-31 questionnaire. The investigators ensured that participants understood all items clearly and answered all parts of the questionnaire.
Further, participants completed the same questionnaire four weeks later with the same method to assess whether there were any changes in their health status.
Data entry and statistical analysis were conducted using Epidata and SPSS version 22.0. Descriptive statistics were used to evaluate the scores’ distribution (mean, range, standard deviation – SD, standard error – SE, median, Interquartile Range – IQR). Internal consistency for each subscale was measured using Cronbach’s alpha coefficient. In this study, Cronbach’s alpha of > 0.6 was considered satisfactory [9]. Test-retest reliability was analyzed via the intra-class correlation coefficient (ICC) with ANOVA test. We chose the model: 2-ways fixed effects, single rater and absolute agreement. ICC values > 0.75, 0.5–0.75, and < 0.5 indicated high, medium, and low reliability, respectively [10]. The correlation between the scores and demographic and clinical characteristics was analyzed using a student’s t test or one-way ANOVA for nominal variables, and Pearson’s correlation coefficient for numeric variables.
3. RESULTS
From a total of 75 eligible PWE, 58 decided to participate in the study from June to December 2020. All participants completed the QOLIE-31 (Figure 2).
In this sample, the mean age (+/- SD) was 37.4 (+/- 13.2) years (range, 18–68). Most participants were married (48.3%), employed (67.2%), had an education of up to high school level (29.3%) and an income below 5 million VND per month (46.5%). Median duration of illness (IQR) was 10 (8, 9) years. Most of the participants experienced focal seizures (67.2%) and had an average seizure per year (48.3%; Table 1).
A summary of descriptive statistics for the QOLIE-31 scores are presented in Table 2. All subscales followed the standards of normal distribution. Mean scores ranged from 55.7 (EF) to 76.4 (SF). Internal consistency and reliability for all subscales, but QOL were satisfactory, with their Cronbach’s alpha ranging from 0.91 to 0.57. Test-retest reliability showed medium to high reproducibility with ICC ranging from 0.72 to 0.87 (p<0.001).
We analyzed construct validity using one-way ANOVA with different demographic and clinical groups and reviewed for statistical significance (p<0.05; Table 3).
(1): Pearson’s correlation coefficicent; (2): Spearman’s correlation coefficicent; (3): ONE-WAY ANOVA; (4): post hoc test showed no statistical difference; (5): post hoc test showed the statistical difference between daily- seizure group and yearly-seizure in SF sub-scale; (6): post hoc test showed the statistical difference between seizure-free for one year group and drug-resistant group in QOL and CF sub-scales
4. DISCUSSION
In this study, internal consistency was satisfactory with a Cronbach’s alpha of > 0.6 in most of the subscales. However, the QOL had the lowest Cronbach’s alpha of 0.57, which was below the alpha of original version [3]. However, the alpha of this subscale in the Spanish, Greek and Turkish version was 0.55, 0.59 and 0.62, respectively [11, 12], which is closer to our translated version. The QOL subscale has only two items, and since the alpha is related to the number of items, this may account for the relatively smaller alpha compared with the other QOLIE-31 scales. Although the authors suggested adding more items in this subscale [11], we believe that the 31-item scale is more appropriate and easier to use in clinical practice. Further, the Cronbach’s alpha of 0.57 is still statistically acceptable [9].
The Vietnamese version showed a test-retest reliability score (ICC from 0.72 to 0.87) that is close to 1.0. In the original scale, the test-retest value ranged from 0.64 to 0.89, and the results from other translations showed a range of 0.60–0.92 [11-16]. These findings suggest that the Vietnamese QOLIE-31 is a reliable measurement tool for PWE.
Regarding the total score of QOLIE-31, the quality of life of PWE was moderate with the mean score of 71.3. Our result was higher than the global mean QOLIE-31 score of 59.8 (SD: 8.0) (n = 7255 people) and 59.7 (8.1) for Southeast Asia [4]. The small sample was one of the reasons for the difference in mean score between Vietnam and other countries; and the other causes could be the dispartiy in culture, expected, social attitude of Vietnamese people and other regions in the world. This difference was also mentioned by Baker when considered among western nations [17]. There were some hypotheses mentioned including differences in societal attitudes, differences in expectations of people with and without epilepsy.
The study’s translational procedure followed the international guideline [7], and reflected the content and construct equivalence of the Vietnamese QOLIE-31 to the original version. Therefore, the validity of Vietnamese version was considered comparable to the original version. In addition, we evaluated the discriminant validity by analyzing the differences of QOLIE-31 total score and subscales among separate groups. Consequently, we found that Seizure Worry was significantly related to Seizure frequency and Cognitive Function was related to deterioration due to age, age of onset, seizure frequency and drug response. Frequent seizures cause the negative effects on the physical, social and psychological well-being of PWE including trauma, sudden death, psychiatric disorders (depression, anxiety), and social stigma. In a multicenter international study with 300 PWE recruited from Germany, United Kingdom, and France, the results highlighted that the seizure types and seizure frequency significantly predicted the quality-of-life scores [17]. The more severe and more frequent seizures were, the lower scores were. Finally, the overall QOLIE-31 score was significantly lower in the drug-resistant group. This hypothesis has been confirmed in other validated versions of the scale [12, 16] and various studies [18, 19].
Notably, the study had several limitations. We were unable to conduct a factor analysis due to the small sample size; further, there is no other available Vietnamese quality of life measurements to test for convergent validity. Furthermore, this current sample was enrolled at the tertiary clinic which might not represent community population. We need to conduct future studies with the larger community sample size.